Maize
Genetics Cooperation Newsletter 80. 2006.
Standardization of a squash technique for endosperm cytology in maize (Zea mays L.)
--Chattopadhyay, K; Sarkar, KR
Standardization of a squash technique for endosperm cytology has been attempted for many years. Different techniques have been proposed from time to time for a suitable cytological methodology for counting the endosperm chromosome number, but a foolproof technique has yet to be developed. The problem is that the endosperm tissue is insensitive to most of the common treatments. Endomitosis and higher chromosome number of endosperm cells in higher ploidy levels sometimes make the chromosome count erroneous. Moreover, higher accumulation of starch in ovules collected from 10 to 12 days after pollination onwards, makes chromosome counting nearly impossible in maize. Early workers (Duncan and Ross, J. Hered. 41:259-268, 1950; Punnett, J. Hered. 44:257-259, 1953) reported a direct squash technique with aceto-carmine or propio-carmine. This technique failed to provide clear cytological pictures. Lin (Stain Technol. 52:197-201, 1977) presented a cytological technique specially suited to maize endosperm. He used 8-hydroxyquinoline, sucrose and aeration to pretreat the tissues. Glusulase enzyme was used to spread the cells along with digestion solution. Endosperm was stained with Feulgen reagent. But during an investigation on the cytogenetic basis of endosperm development, the protocol given by Lin failed to give good results in our laboratory. The aim of the present cytological study was to standardize a reliable protocol or to make modifications to improve Lin�s squash technique for endosperm cytology in maize.
The materials used in this study included two diploid inbred lines (CM105 and CM111), one tetraploid line (a derivative of Alexander�s Synthetic B tetraploid), two sets of triploid lines (obtained by crossing the tetraploid stock by two diploid inbred lines, CM105 and CM111) and one trisomic stock (primary trisomy for chromosome 10) of maize. Selfing and inter-ploidy crosses were made and immature ear shoots were collected from the field at one to eight days after pollination. The shoots were fixed in 3:1 alcohol : acetic acid after pretreating with 7.5% sucrose (granular) and 0.03% 8-hydroxyquinoline for 4-6 hours.
In the present study, different protocols reported for maize were tried and the following procedure was found most convenient and reliable for counting chromosomes from endosperm cells.
Fixed ovules were washed with 70% alcohol. The external coat of lemma, palea, glumes and pericarp were removed. The whitish portion of the ovule containing the embryo sac was squeezed out of the ovule, collected in a vial and subjected to the following procedure:
a. Ovule segments were transferred through 75%, 50% and 30% alcohol solutions for gradual hydration and then washed with distilled water.
b. The material was treated with 1N HCl for 8 minutes at 60oC. Prolonged treatment caused swelling of chromosomes. Washing with distilled water followed.
c. Feulgen staining was carried out for a long period for better staining (6 hours), keeping the vials in a dark place at 10-15o C.
d. Stained ovules were washed with distilled water for 10-15 minutes and then with 50% acetic acid for 4-5 minutes to remove the excess stain.
e. Enzyme treatment: 3% pectinase (CDH) solution for 6-8 hours or a solution containing 2.5% pectinase (CDH) and 2.5% cellulase (Merck) for half an hour was found to be very effective for softening the cell wall and maceration.
f. After removal of the enzyme solution, ovules were washed with acetic acid (50%) for 5-10 minutes for further softening of the tissues.
g. One ovule, or part of it, was then placed on a slide and squashed with one drop of 1% aceto-carmine stain for 4-5 minutes. A cover slip was then placed on top and sealed with paraffin wax.
h. To make the slide permanent, it was placed in 45% acetic acid vapor for one to two days. After removal of the paraffin wax seal, the slide was placed in a 1:1 acetic acid and Butan-1-ol mixture. When the cover slip detached from the slide, the slide was transferred to Butan-1-ol for 3-4 minutes. After drying, one drop of DPX was added and the cover slip was placed over it. Chromosome numbers were counted from dividing (metaphase stage) endosperm cells and photographs were taken with a Nikon Microphot FX.
Some of the results obtained by using this technique are shown in Figures 1 and 2. By using this technique, chromosome counting in endosperm cells of higher ploidy levels was not entirely accurate in each and every squash preparation because of overcrowding and poor spread of chromosomes. More hand pressure and heating sometimes improved the spreading of metaphase chromosomes of the endosperm cells.
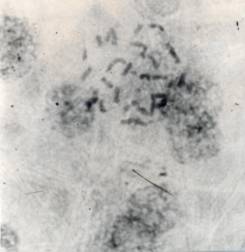
Figure 1. Endosperm cell with 30 chromosomes from the ovule of a
diploid (2n) and diploid (2n) cross, collected after 6 days of pollination.

Figure 2. Endosperm
cell with 30 chromosomes from the ovule of a trisomic (2n+1) and diploid (2n)
cross, collected 5 days after pollination.
_________________________________________________
Please
note:
Notes submitted to the Maize Genetics Cooperation Newsletter may be cited only
with consent of the authors.
Return to MNL 80 on-line index.
Return to MNL index.
Return to MaizeGDB home page
____________________________________________