Maize Genetics Cooperation Newsletter vol 84 2010
Please Note: Notes submitted to the Maize Genetics Cooperation
Newsletter may be cited only with consent of authors.
TALLAHASSEE, FLORIDA
Florida State University
A recombinant
C-terminal fragment of maize Initiator Binding Protein 2 (IBP2) binds to
telomere-repeat DNA in vitro.
Moore, JM
Bass, HW
The
maize (Zea mays L.) initiator-binding protein2 (IBP2) gene is known to encode a protein,
IBP2, that binds to the initiator
(inr)
element in the Shrunken1 promoter
(Lugert and Werr 1994; Plant
Mol Biol, 25, 493–506). The duplicate genes IBP1 and IBP2 were
initially identified in ligand-binding screens using cDNA expression libraries. The binding of IBP to the transcription start site inr element was
confirmed by footprint and band-shift assays. We and others have observed that
IBP1 and IBP2 also resemble telomere DNA-binding proteins from other plant
species. We investigated therefore
whether the IBP2 protein exhibits telomere DNA-binding activity.
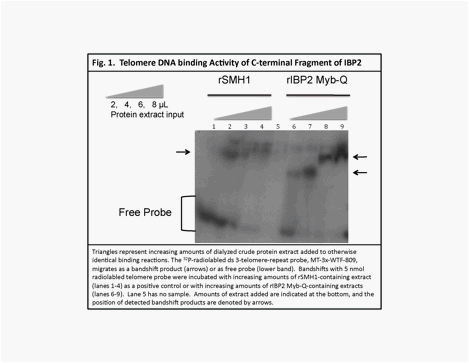
Using
Electrophoretic Mobility Shift Assay (EMSA) we found
that a C-terminal sublcone of the IBP2 protein (rIBP2
Myb-Q, AA 579-667, GenBank GU080214),
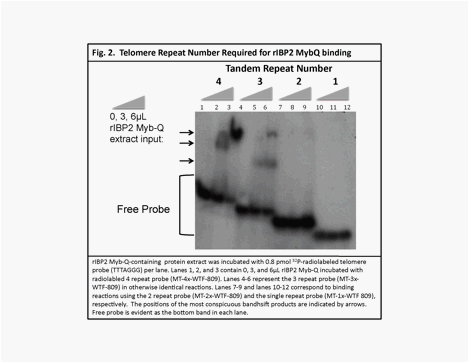
binds telomeric repeat DNA in vitro (Fig. 1, arrows indicate band shifts). We also found that rIBP2 Myb-Q binds strongly to double stranded telomere repeat
sequences with three or four tandem repeats, but only weakly to sequences with
two (Fig. 2, arrows indicate band shifts). Furthermore, we found no evidence of binding with only one
telomere repeat (5� TTTAGGG 3�) sequence. Single point mutations (T1:A and G6:T) in the three-repeat sequence diminished, but
did not entirely abolish, binding activity (via competition EMSA assays, data
not shown).
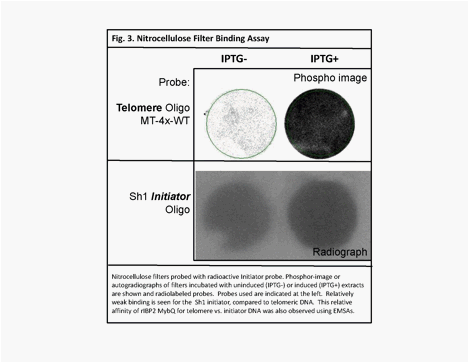
In
order to compare the binding of rIBP2 Myb-Q to
telomere vs. initiator DNA, we carried out filter binding assays. In this experiment, total protein
extract from E coli cultures expressing rIBP2 Myb-Q
(IPTG+) were incubated with nitrocellulose filters. These filters were then incubated with radiolabeled
oligonucleotide probes corresponding to telomere DNA
or Sh1 initiator DNA (Fig. 3).
From these experiments, we found that the rIBP2 Myb-Q
showed very weak binding to the initiator
sequence of the Shrunken1 promoter. These findings confirm the prediction
that the maize IBP protein (IBP1 or IBP2) has telomere-DNA-binding
activity. Consequently, maize
IBP1/2 may reside at maize telomeres, raising the intriguing possibility of
dual functions for IBP, telomeric and
transcriptional.